Mirati's (MRTX) Stock Rallies 11% on Buyout Speculations
Mirati Therapeutics MRTX stock was up 11.3% on Wednesday on reports that big cancer drugmakers may offer to buy the company ahead of some upcoming important company milestones.
However, in the year so far, the stock price of Mirati has plunged 34.7% compared with the industry’s decline of 19.5%.
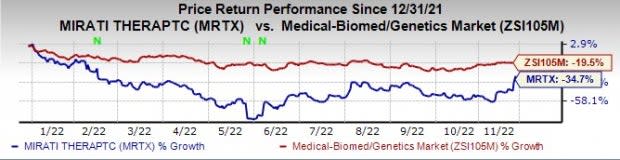
Image Source: Zacks Investment Research
Mirati’s new drug application (NDA), seeking approval for adagrasib, its KRAS inhibitor drug, for previously-treated KRASG12C-mutated non-small cell lung cancer (NSCLC) is under review with the FDA. A decision by the FDA is expected by Dec 14, 2022. A marketing authorization application (MAA) is also under review in Europe. Both the NDA and the MAA are based on data from the phase II registration-enabling cohort of the KRYSTAL-1 study. If adagrasib is approved and can outperform any potential competitor from the same class, it will be a huge boost to the stock. This is the primary reason for attracting promising buyout offers.
Potential buyers could be Pfizer, Bristol-Myers BMY, Merck MRK or AstraZeneca, per an article on Investor's Business Daily. Mirati did not issue any press release or make any comment on the buyout speculations. There are currently no formal offers and a deal is not imminent.
Mirati is also evaluating adagrasib — both as monotherapy and combinations — in multiple cohorts of phase I/II KRYSTAL-1 study across multiple solid tumors that harbor KRAS G12C mutations. These include a combination of adagrasib with Merck’s Keytruda in NSCLC, adagrasib plus Boehringer Ingelheim’s Gilotrif (afatinib) in advanced NSCLC and adagrasib with Bristol-Myers’ Erbitux (cetuximab) in advanced colorectal cancer or CRC. Top-line interim data from another study, KRYSTAL-7 study evaluating adagrasib plus Merck’s Keytruda in first-line NSCLC is expected to be released in the fourth quarter. If the data are positive, it can further increase interest in Mirati.
Adagrasib, if approved, will face competition from Amgen’s AMGN KRAS G12C inhibitor, Lumakras, which was approved for second-line NSCLC in the United States in 2021 and in Europe in January 2022. It is now launched in more than 45 countries. Amgen’s Lumakras is off to an excellent start, while its label expansion studies, which have the potential to significantly expand the currently addressable patient population, are progressing rapidly.
Mirati’s other pipeline candidate, sitravatinib, is being evaluated in a pivotal phase III study (SAPPHIRE) combined with Bristol-Myers’ Opdivo for second-line or third-line non-squamous NSCLC. Interim data from the study is expected in the fourth quarter of 2022. If the outcome turns out to be positive, it could be the basis of regulatory submissions for sitravatinib in the United States and Europe, potentially by mid-2023.
Zacks Rank
Mirati currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report
Amgen Inc. (AMGN) : Free Stock Analysis Report
Mirati Therapeutics, Inc. (MRTX) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 