Biotech Stock Roundup: BBIO, ARGX Soar on Study Results, APLS Down on Safety Issues
It was a busy week for the biotech sector, with many important regulatory and pipeline updates. Among these, key news was the success of BridgeBio’s BBIO’s late-stage study.
Recap of the Week’s Most Important Stories:
BridgeBio Surges on Study Results: Shares of BridgeBio surged after the company announced that the phase III study of acoramidis in transthyretin amyloid cardiomyopathy ( ATTR-CM) achieved its primary goal. The late-stage study, ATTRibute-CM, was designed to study the efficacy and safety of acoramidis, an investigational, next-generation, orally-administered, highly potent, small molecule stabilizer of transthyretin (TTR).
The study's primary endpoint was determined through a hierarchical analysis (prioritizing in order) - all-cause mortality, the frequency of cardiovascular-related hospitalization, change from baseline in NT-proBNP and change from baseline in six-minute walk distance. Results showed a highly statistically significant improvement in the primary endpoint demonstrated by a win ratio of 1.8.
Eight percent of ties in Finkelstein-Schoenfeld (F-S) primary analysis broken by all-cause mortality and frequency of cardiovascular-related hospitalization and statistical significance was also achieved on an F-S test in those two parameters alone. After 30 months, 81% survival rate among patients was observed who received acoramidis compared with 74% of those in the placebo group. In addition, there was a significant relative risk reduction of 50% in the frequency of cardiovascular-related hospitalizations.
BridgeBio intends to submit its NDA to the FDA before the end of 2023, with regulatory filings in additional markets to follow in 2024.
Apellis Crashes on Safety Issues: Shares of Apellis Pharmaceuticals, Inc. APLS nosedived after the company received reports of retinal vasculitis (or inflammation) following treatment with Syfovre (pegcetacoplan injection). The FDA approved Syfovre for treating geographic atrophy (GA) secondary to age-related macular degeneration (AMD) in February. The American Society of Retina Specialists Research and Safety in Therapeutics (ReST) Committee collaborated with Apellis Pharmaceuticals to investigate the reported retinal vasculitis events. Out of the six reported cases, two were confirmed as occlusive, one as non-occlusive and the remaining three were undetermined due to limited information. These events occurred seven and 13 days after the initial administration of Syfovre, with no specific lots implicated.
The company, in close coordination with external experts and the ReST Committee, is conducting a thorough investigation into each event. Apellis Pharmaceuticals has followed up with the FDA and provided detailed information on these cases. The FDA has not recommended any immediate action in response to the reports. The reported vasculitis events occurred at an estimated rate of 0.01% per injection, or approximately 1 in 10,000 injections. Per the company, about 60,000 vials of Syfovre have been distributed since approval. Apellis stated that there were no instances of retinal vasculitis reported during the clinical trials, which involved more than 23,000 injections.
Apellis currently carries a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Vir Down on Pipeline Update: Shares of Vir Biotechnology, Inc. VIR lost 44.90% after the company announced that its phase II PENINSULA (PrevEntioN of IllNesS DUe to InfLuenza A) study evaluating VIR-2482 for the prevention of symptomatic influenza A illness did not meet primary or secondary efficacy endpoints. The study is the first phase II outpatient trial to evaluate the role of a monoclonal antibody in the prevention of influenza A illness. The dose-ranging, proof-of-concept trial enrolled approximately 3,000 men and women aged 18 to 64 without risk factors for serious complications from an influenza infection who did not receive an influenza vaccination for the flu season.
Data showed that the participants who received the highest dose of VIR-2482 (1,200 mg) experienced a reduction of approximately 16% in influenza A protocol-defined illness, which was statistically not significant. Participants who received the highest dose showed an approximately 57% reduction in symptomatic influenza A illness when defined according to CDC influenza-like-illness criteria, which was one of two secondary endpoints. Nevertheless, the candidate was generally well tolerated and no safety signals were identified.
Vir Biotech is now focused on advancing next-generation solutions for serious respiratory infections, including VIR-2981, an investigational neuraminidase-targeting monoclonal antibody against influenza A and B viruses.
argenx Gains on Study Results: Shares of argenx SE ARGX gained after the company announced positive top-line results from the ADHERE study. The study is evaluating Vyvgart Hytrulo (efgartigimod alfa and hyaluronidase-qvfc) in adults with chronic inflammatory demyelinating polyneuropathy (CIDP). ADHERE enrolled adults who were treatment naïve (not on active treatment within the past six months) or currently on immunoglobulin therapy or corticosteroids. The trial consisted of a run-in period where current treatment was stopped, followed by an open-label Stage A, after which responders to Vyvgart Hytrulo advanced to a randomized, placebo-controlled Stage B.
The study met its primary endpoint, demonstrating a significantly lower risk of relapse with Vyvgart Hytrulo compared to placebo. Vyvgart Hytrulo demonstrated a 61% reduction in risk of relapse versus placebo. Of eligible patients, 91% continued to the ADHERE-Plus open-label extension study. Zai Lab has an exclusive license agreement with argenx for the development and commercialization of Vyvgart and Vyvgart Hytrulo in Greater China.
Performance
The Nasdaq Biotechnology Index has gained 0.91% in the past five trading sessions. Among the biotech giants, Gilead has gained 4.21% during the period. Over the past six months, shares of MRNA have lost 35.68%. (See the last biotech stock roundup here: Biotech Stock Roundup: BIIB, Eisais Drug Update, NVAX Gains on Deal Amendment).
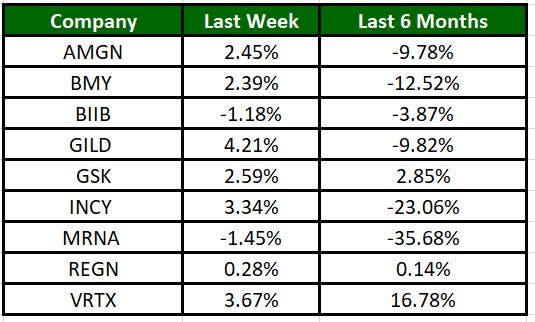
Image Source: Zacks Investment Research
What's Next in Biotech?
Stay tuned for earnings and more pipeline updates.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
argenex SE (ARGX) : Free Stock Analysis Report
Apellis Pharmaceuticals, Inc. (APLS) : Free Stock Analysis Report
BridgeBio Pharma, Inc. (BBIO) : Free Stock Analysis Report
Vir Biotechnology, Inc. (VIR) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 