Sanofi, Regeneron's Dupixent Gets FDA Nod for 4th Indication
Sanofi SNY and partner Regeneron REGN announced that the FDA has approved its blockbuster medicine Dupixent (dupilumab) for a new indication — eosinophilic esophagitis (EoE).
The FDA has approved the use of Dupixent for treating adults and children aged 12 and older with EoE, a chronic, progressive inflammatory disease. The FDA’s decision came much earlier than the PDUFA date of Aug 3, 2022. Dupixent is the first and only medicine indicated for eosinophilic esophagitis in the United States.
EoE inflates and damages the esophagus, which makes it difficult for the patients to eat and swallow, leading to poor quality of life. There are no approved treatments specifically for this disease, thus creating huge unmet demand. Dupixent, in studies, has demonstrated that it improves the ability to swallow and also reduce the inflammation in the esophagus.
EoE is the fourth disease for which Dupixent is now indicated and also represents the first indication for a gastrointestinal disease. Sanofi and Regeneron’s Dupixent is already approved in the United States, EU and some other countries for three type II inflammatory diseases, namely severe chronic rhinosinusitis with nasal polyposis, severe asthma and moderate-to-severe atopic dermatitis in different age populations. Dupixent is under review in Europe for EoE.
Sanofi stock has risen 8.6% this year so far compared with the industry’s increase of 4.4%.
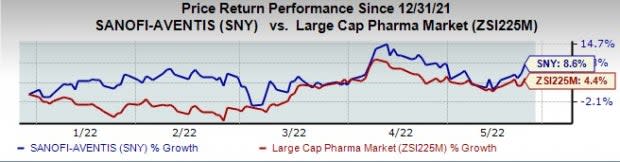
Image Source: Zacks Investment Research
Dupixent is being jointly marketed by Regeneron and Sanofi under a global collaboration agreement. Sanofi records global net product sales of Dupixent while Regeneron records its share of profits/losses in connection with the global sales of the drug.
Dupixent sales are now annualizing at close to €6.0 billion after around three years on the market. Sanofi expects Dupixent to achieve more than €13 billion in peak sales. The drug’s frequent label expansion approvals are driving sales higher. With outside U.S. revenues accelerating and multiple approvals for new indications and expansion in younger patient populations expected in the near future, its sales are expected to be higher
Dupixent has become the key driver of the top line for Sanofi. Dupixent generated first-quarter global product sales of $1.61 billion, which were recorded by Sanofi.
Sanofi and Regeneron are also studying dupilumab in late-stage studies in a broad range of diseases, driven by type II inflammation like bullous pemphigoid, chronic spontaneous urticaria, prurigo nodularis, and chronic obstructive pulmonary disease.
In a separate press release, Sanofi announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has given a positive opinion recommending the approval of Xenpozyme (olipudase alfa) for non-CNS clinical manifestations of acid sphingomyelinase deficiency (ASMD) disease. Xenpozyme is already approved in Japan for ASMD, a rare and life-threatening disease. The CHMP opinion was based on data from two clinical studies, in which Xenpozyme led to robust and clinically relevant improvement in lung function and reduced spleen and liver volumes. The European Commission is expected to give its decision in a few months.
Sanofi currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Stocks to Consider
Some better-ranked biotech stocks are Alkermes ALKS and Sesen Bio SESN, both with a Zacks Rank #1.
The Zacks Consensus Estimate for Alkermes’ 2022 loss per share has narrowed from 13 cents to 3 cents in the past 30 days. Shares of ALKS have risen 29.0% year to date.
Earnings of Alkermes beat estimates in each of the last four quarters, the average being 350.5%.
The Zacks Consensus Estimate for Sesen Bio’s 2022 loss has declined from 33 cents to 32 cents per share in the past 30 days. Shares of SESN have declined 39.4% in the year-to-date period.
Earnings of Sesen Bio beat estimates in three of the last four quarters and missed the mark on one occasion, the average surprise being 69.9%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Regeneron Pharmaceuticals, Inc. (REGN) : Free Stock Analysis Report
Sanofi (SNY) : Free Stock Analysis Report
Alkermes plc (ALKS) : Free Stock Analysis Report
SESEN BIO, INC. (SESN) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 