Karuna (KRTX) Stock Skyrockets 98.7% in a Month: Here's Why
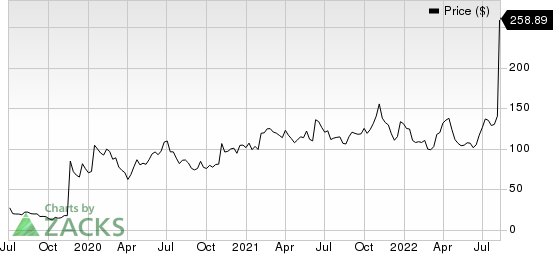
Shares of Karuna Therapeutics KRTX have surged 98.7% in the past month against the industry’s 1.6% decline.
One of the main reasons for this rise in stock price is management’s announcement of positive topline results from the phase III EMERGENT-2 study evaluating KarXT, its lead investigational therapy, in adults with schizophrenia earlier this month. The study met its primary and key secondary endpoints.
KarXT exhibited a statistically significant and clinically meaningful 9.6-point reduction in the Positive and Negative Syndrome Scale (PANSS) total score over placebo, thereby meeting the study’s primary endpoint. PANSS is a scale for measuring schizophrenia symptom severity.
The EMERGENT-2 study also met its secondary endpoint as KarXT demonstrated an early and sustained statistically significant reduction in both positive symptoms (like hallucinations or delusions) and negative symptoms (like, difficulty enjoying life or withdrawal from others) of schizophrenia. The drug’s safety profile was consistent with previously conducted clinical studies.
Unlike current standard-of-care antipsychotic treatments, which primarily work by inhibiting dopamine and serotonin receptors while also carrying many debilitating side effects, KarXT works by preferentially stimulating M1 and M4 muscarinic receptors in CNS. A stimulation in these receptors has demonstrated improvements in psychosis and cognition.
The EMERGENT-2 study is a part of the broader EMERGENT clinical program, which is evaluating KarXT for treating acute psychosis in adults with schizophrenia. The program also consists of one completed phase II study (EMERGENT-1) and three ongoing late-stage studies, namely EMERGENT-3, EMERGENT-4 and EMERGENT-5, evaluating the efficacy and long-term safety of KarXT in adults with schizophrenia.
Topline data from the EMERGENT-3 study is expected in first-quarter 2023. Based on data from the EMERGENT program, Karuna expects to file a new drug application (NDA) with the FDA for KarXT as a treatment for schizophrenia in mid-2023.
Given the potential of KarXT over the existing standard-of-care therapies, Karuna Therapeuticsis also evaluating the candidate in the phase III ARISE study as an adjunctive treatment of schizophrenia in adults who experience an inadequate response to their current antipsychotic therapy. While KRTX is currently enrolling patients in this study, topline data is expected in first-half 2024.
Apart from schizophrenia, Karuna Therapeutics is developing KarXT as a potential treatment of dementia-related psychosis (DRP). KRTX is initially focusing on developing KarXT to treat psychosis in AD, one of the most prevalent subtypes of DRP. KRTX remains on track to start the phase III ADEPT clinical program, evaluating the candidate for psychosis in elderly patients with Alzheimer’s disease (AD). This program will consist of three late-stage studies, with the first expected to start in third-quarter 2022 and the remaining studies in the next year.
Apart from AD, KRTX has also shown interest in other DRP subtypes and intends to start development studies in the future.
KarXT is consists of two components — xanomeline, a novel muscarinic agonist acting as an antipsychotic and procognitive therapeutic agent; and trospium, a muscarinic antagonist to limit side-effects of xanomeline.
We note that xanomeline is a novel muscarinic agonist initially developed by Eli Lilly LLY, which was licensed to Karuna in 2012. Per the terms of licensing deal, Karuna is obligated to pay potential regulatory and development milestone payments to Eli Lilly of up to $70 million. In addition, Eli Lilly is also eligible to receive tiered royalties, ranging from low to mid-single-digit percentages, on the future net sales of the drug.
Karuna Therapeutics, Inc. Price
Karuna Therapeutics, Inc. price | Karuna Therapeutics, Inc. Quote
Zacks Rank & Stocks to Consider
Karuna Therapeutics currently carries a Zacks Rank #3 (Hold). Some better-ranked stocks in the overall healthcare sector are Geron GERN and Morphic MORF. While Morphic sports a Zacks Rank #1 (Strong Buy) at present, Geron carries a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 30 days, estimates for Morphic’s 2022 loss per share have narrowed from $3.47 to $1.75. Loss estimates for 2023 have narrowed from $3.96 to $3.77 during the same period. Shares of Morphic have lost 29.6% in the year-to-date period.
Earnings of Morphic beat estimates in three of the last four quarters and missed the mark just once, witnessing a surprise of 48.29%, on average. In the last reported quarter, MORF delivered an earnings surprise of 183.95%.
In the past 30 days, estimates for Geron’s 2022 loss per share have narrowed from 38 cents to 36 cents. Loss estimates for 2023 have narrowed from 36 cents to 35 cents during the same period. Shares of Geron have surged 77.9% in the year-to-date period.
Earnings of Geron beat estimates in three of the last four quarters and missed the mark just once, witnessing a surprise of 1.07%, on average. In the last reported quarter, GERN delivered an earnings surprise of 18.18%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Geron Corporation (GERN) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report
Morphic Holding, Inc. (MORF) : Free Stock Analysis Report
Karuna Therapeutics, Inc. (KRTX) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 