GSK NDA for Myelofibrosis Candidate Gets FDA Acceptance
GSK plc GSK announced that the FDA has accepted its new drug application (NDA) seeking approval for momelotinib for treating myelofibrosis, a fatal cancer of the bone marrow. The FDA granted standard review to the NDA and so its decision is expected on Jun 16, 2023.
Momelotinib was added to GSK’s hematology portfolio with the July acquisition of California-based cancer biotech, Sierra Oncology.
GSK’s stock has declined 37.1% this year so far compared with a decrease of 17.7% for the industry.
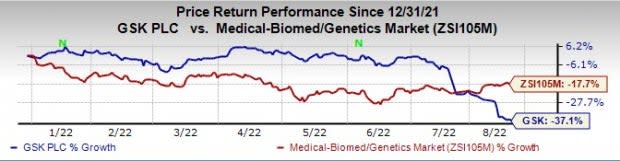
Image Source: Zacks Investment Research
The NDA filing was based on data from key phase III studies, including the pivotal MOMENTUM study. The MOMENTUM study evaluated momelotinib versus danazol for treating anemic myelofibrosis patients previously treated with an approved JAK inhibitor.
Myelofibrosis disease often leads to anemia that causes fatigue, increased risk of infection and bleeding or bruising due to reduced platelet count in patients. In the MOMENTUM study, momelotinib achieved statistical significance in the primary and all pre-specified secondary endpoints, reporting a statistically significant benefit on symptoms, anemia and splenic size.
Momelotinib particularly complements GSK’s new drug, anti-BCMA therapy, Blenrep/belantamab mafatotin, which was approved for fourth-line multiple myeloma in 2020. Blenrep is being studied in phase III for third-line multiple myeloma.
Zacks Rank & Stocks to Consider
GSK currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Some better-ranked stocks in the same sector are Alkermes ALKS, Editas Medicine EDIT and Immunovant IMVT, all carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Alkermes’ estimates for 2022 have improved from a loss of 16 cents to earnings of 20 cents over the past 30 days. Earnings estimates for 2023 have gone up from 32 cents to 50 cents over the same time frame. Shares of Alkermes have risen 10.1% this year so far.
Earnings of Alkermes beat estimates in all the last four quarters, delivering a surprise of 325.48%, on average.
Editas Medicine’ loss per share estimates for 2022 have narrowed from $3.41 to $3.35 in the past 30 days. EDIT’s stock is down 38.6% in the year-to-date period.
Earnings of Editas Medicine’ beat estimates in all the last four quarters, the average surprise being 17.82%.
Immunovant’s stock is down 41.5% this year so far. Loss estimates for 2022 have narrowed from $1.66 per share to $1.64 per share while that for 2023 have improved from $1.94 per share to $1.75 per share over the past 30 days.
Earnings of Immunovant missed estimates in three of the last four quarters and beat the mark once, the average negative surprise being 8.97%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
GSK PLC Sponsored ADR (GSK) : Free Stock Analysis Report
Alkermes plc (ALKS) : Free Stock Analysis Report
Editas Medicine, Inc. (EDIT) : Free Stock Analysis Report
Immunovant, Inc. (IMVT) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 