Regeneron's (REGN) Eylea Gets FDA Nod for ROP in Infants
Regeneron Pharmaceuticals, Inc. REGN announced that the FDA has approved the label expansion of ophthalmology drug Eylea (aflibercept).
Eylea injection has been approved for the treatment of preterm infants with retinopathy of prematurity (ROP).
The FDA’s approval is supported by data from two randomized global phase III studies – FIREFLEYE (n=113) and BUTTERFLEYE (n=120) – evaluating Eylea 0.4 mg versus laser photocoagulation (laser) in infants with ROP.
Data from both trials showed that approximately 80% of Eylea-treated infants achieved an absence of both active ROP and unfavorable structural outcomes at 52 weeks of age, which is better than would have been expected without treatment.
Both trials were conducted pursuant to FDA Pediatric Written Request. In October 2022, the FDA granted pediatric exclusivity to Eylea, which extends the period of U.S. market exclusivity of the drug by an additional six months through May 17, 2024.
Eylea is now indicated to treat five retinal conditions caused by ocular angiogenesis following the first pediatric approval.
The drug is approved for the treatment of patients with wet age-related macular degeneration (AMD), macular edema following retinal vein occlusion (RVO), diabetic macular edema (DME), diabetic retinopathy (DR) and ROP.
Regeneron is developing Eylea in collaboration with Bayer BAYRY.
Regeneron sponsored BUTTERFLEYE, while Bayer sponsored FIREFLEYE.
Regeneron records net product sales of Eylea in the United States and Bayer records net product sales of Eylea outside the United States. Regeneron records its share of profits/losses in connection with sales of Eylea outside the United States.
Regeneron’s shares have gained 20.1% in the past year against the industry’s decline of 12.8%.
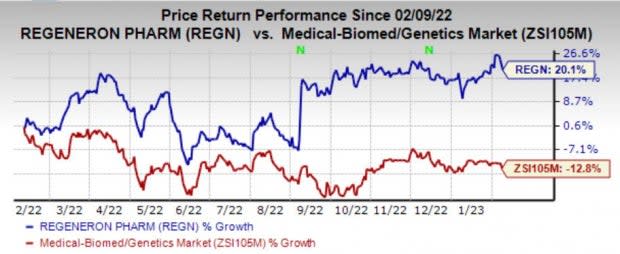
Image Source: Zacks Investment Research
Regeneron delivered better-than-expected fourth-quarter 2022 results even though Eylea sales declined.
Meanwhile, the primary endpoints were achieved in two trials investigating aflibercept 8 mg with 12- and 16-week dosing regimens in patients with DME and wet AMD. The PHOTON trial in DME and the PULSAR trial in wet AMD both demonstrated that aflibercept 8 mg 12- and 16-week dosing regimens achieved non-inferiority in vision gains compared to the Eylea 8-week dosing regimen.
A biologics license application (BLA) for the treatment of wet AMD and DME was submitted to the FDA in December 2022.
Meanwhile, shares of Bayer rallied after the company appointed Bill Anderson as its CEO. Anderson had recently served as CEO of Roche’s Pharmaceuticals Division.
Zacks Rank and Key Picks
Regeneron currently carries a Zacks Rank #3 (Hold). A couple of better-ranked stocks in the biotech sector are Syndax Pharmaceuticals SNDX and Dynavax Technologies DVAX. While Syndax sports a Zacks Rank #1 (Strong Buy), Dynavax carries a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, loss estimates for Syndax for 2022 have narrowed to $2.48 from $2.53. Syndax also surpassed estimates in each of the trailing four quarters, with the average surprise being 95.39%.
Over the past 60 days, earnings estimate for Dynavax for 2022 have increased by 11 cents to $1.95. Dynavax also surpassed estimates in two of the trailing four quarters, with the average surprise being 73.15%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Regeneron Pharmaceuticals, Inc. (REGN) : Free Stock Analysis Report
Dynavax Technologies Corporation (DVAX) : Free Stock Analysis Report
Bayer Aktiengesellschaft (BAYRY) : Free Stock Analysis Report
Syndax Pharmaceuticals, Inc. (SNDX) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 